SAN FRANCISCO, April 29, 2026 (GLOBE NEWSWIRE) -- Laguna Biotherapeutics, Inc. (Laguna), a clinical-stage biotechnology company focused on novel live bacterial therapeutics, today announced that the U.S. Food and Drug Administration (FDA) has cleared its Investigational New Drug (IND) application, granting a "safe to proceed" for its lead clinical candidate from the QUAIL platform, LGNA-100.
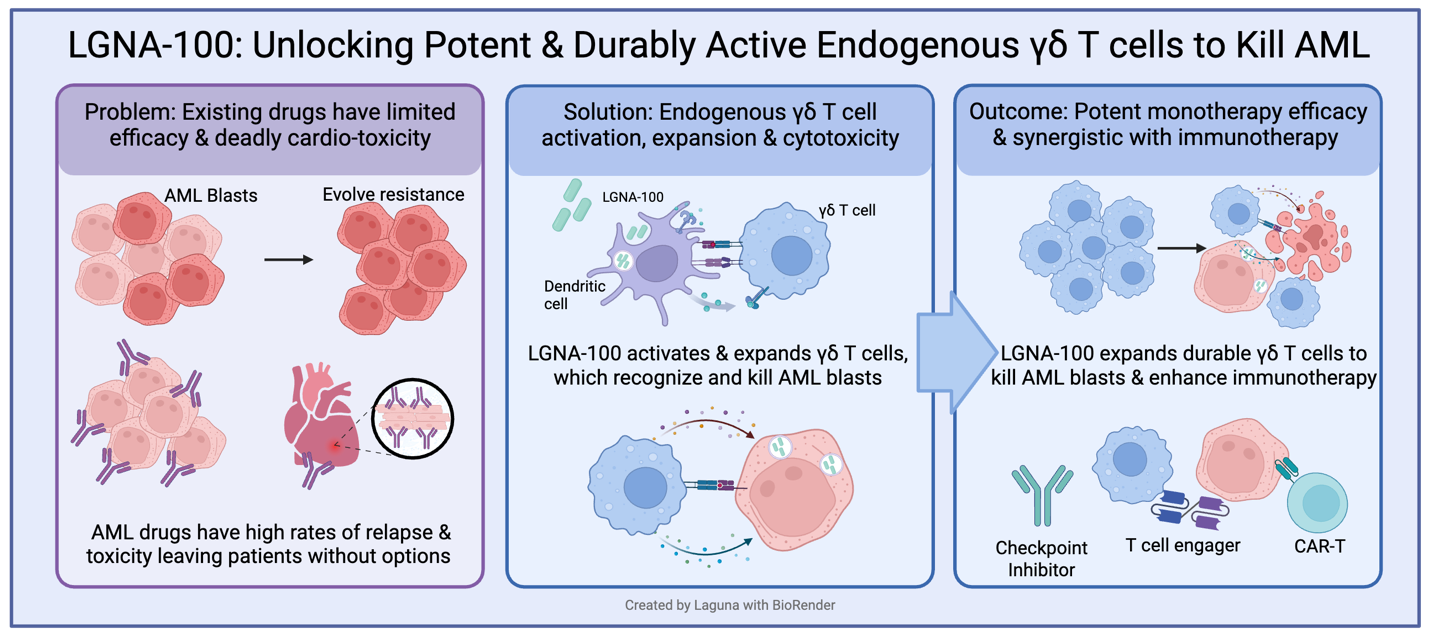
LGNA-100 is a first-in-class, attenuated live bacterial immunotherapy designed to safely harness our immune system's evolved response to Listeria; robustly and durably expanding and activating endogenous γδ T cells that can directly kill cancer cells while also improving existing immunotherapies. The Phase 1 first-in-human study will validate the QUAIL platform and evaluate LGNA-100 in patients with high-risk leukemia following hematopoietic stem cell transplantation (HSCT) to prevent leukemic relapse.
"The IND clearance of our first clinical study is a defining moment and transformative milestone for Laguna as we transition into a clinical-stage company," said Jonathan Kotula, Ph.D., CEO of Laguna. "Our goal is to create systems-level therapies for complex diseases. With LGNA-100, and the QUAIL platform we are taking a fundamentally new approach to selectively stimulate innate T cells to improve long-term outcomes for pediatric patients with high-risk leukemia."
The clinical rationale for the QUAIL platform builds directly upon decades of research into the human γδ T cell response to Listeria, and the protective role of γδ T cells against leukemia recurrence following HSCT. A presentation covering our clinical rationale will be presented at the ISCT 2026 Annual Meeting on May 6 in Dublin, Ireland.
"In the setting of αβ-depleted HSCT, γδ T cells are critical effectors that provide potent graft-versus-leukemia activity without driving graft-versus-host disease," said Dr. Alice Bertaina, MD, PhD, Co-Director of the Bass Center for Childhood Cancer and Blood Diseases, Lucile Packard Children's Hospital at Stanford University and lead Clinical Advisor for Laguna. "While early pharmacologic activators like zoledronic acid (ZOL) showed the clinical potential of this approach, intense ZOL stimulation pushes these cells into a more mature, terminally differentiated and exhausted state. Our comprehensive preclinical evaluations demonstrate that LGNA-100 drives a distinct, multifunctional γδ T cell response with improved kinetic, phenotypic, and functional features compared to ZOL, supporting more durable activation without hyperactivation or early exhaustion. I am very excited to see this translated into the clinic for these high-risk leukemia patients."
"Pediatric AML remains one of the most challenging frontiers in oncology, demanding novel modalities that can detect and kill the disease without compounding toxicities," said Bill Newell, former CEO of Sutro Biopharma and Strategic Advisor to Laguna. "Having spent years evaluating platforms to tackle these exact malignancies, I believe Laguna's approach using γδ T cells to potentially solve the problems associated with high-risk leukemia is a massive leap forward. Securing this IND is a testament to the rigor of their science and positions LGNA-100 as a highly differentiated asset in the cancer immunotherapy space."
The Phase 1 clinical study is a company-sponsored open-label, first-in-human, single ascending dose study designed to assess safety and tolerability, and support the proof of LGNA-100's mechanism of action. The study will evaluate LGNA-100 administered via intravenous (IV) infusion in pediatric and young adult participants with high-risk acute leukemias and MDS who have received an αβ-depleted HSCT.
About LGNA-100
LGNA-100 also known as QUAIL-100 is an investigational cancer immunotherapeutic agent derived from Listeria monocytogenes (Lm), developed from the QUAIL platform, designed to activate and expand a patient's endogenous γδ T cells.
About Laguna Biotherapeutics, Inc. (Laguna)
Laguna is a privately funded, clinical-stage biotechnology company headquartered in San Francisco, CA with research labs in Siena, Italy. It is focused on developing novel therapeutics that leverage Lm to activate and expand the immune system to create impactful treatments across oncology, as well as inflammatory and infectious disease.
A photo accompanying this announcement is available at https://www.globenewswire.com/NewsRoom/AttachmentNg/14da867a-1402-4d3d-b473-d3267f862b39
-
微创再突破!北京安贞医院完成北京市首例蛇形臂单孔机器人经肋间肺叶切除术近日,北京安贞医院胸外科迎来微创外科领域的又一重大突破。在科室主任游宾的指导下,副主任医师郭琳主刀,医师文韬为助手,成功完成北京地区首例蛇形臂单孔机器人经肋间2026-04-30
-
AI赋能教育更有温度!第87届教装展,海亮科技解锁未来教育新模样AI赋能教育更有温度!第87届教装展,海亮科技解锁未来教育新模样2026-04-30
-
风从东方来|大国茶韵京东自营 盛启全国八仓速达风从东方来京东自营盛大启幕, 全国速达,静候时代品味者共赏东方本味。 风从东方来,中国原叶汽泡茶开创者。 承东方茶脉,融当代新质,以匠心铸品质2026-04-30
-
康道,做好灵芝 共筑健康美好生活在这个连养生都追求“速效”的时代,有家企业却像一股清流,在大别山深处一扎就是40年。别人追风口,它追的却是灵芝。它就是康道。它的slogan很朴素——“康道,做好2026-04-30
-
菜百股份发布2025年年报,营收、净利润实现双位数增长2025年国际金价频频刷新历史新高,全国限额以上单位金银珠宝零售额达3,736亿元,同比增长12.8%,行业消费呈现复苏与回暖。 4月29日,北京菜市口百货股份有限公2026-04-30
-
AMD股价暴跌17%创近9年之最,苏姿丰紧急回应:AI增速远超想象
-
江苏省脑机接口产业联盟在宁成立,麦澜德分享前沿成果
-
艾芬达入选国家知识产权强国建设示范创建对象:二十载长期主义,兑现每一份用户价值
-
慧启赣疆 聚势共赢丨慧友酒店集团江西品鉴会书写区域文旅融合新篇
-
电影《一秒》定档:2026年,活在这一秒
-
西藏斜视患儿寒假进京手术成功,千里护航点亮视觉未来
-
年度盛典|卓兴半导体2025年度总结表彰暨 2026 年迎新晚会
-
倒计时21天!2026未来医疗医药100强大会议程再刷新
-
科技赋能 生态协同,登途集团车辆资产管理运营模式助推行业提质增效
-
公元地暖构建“产品+施工+服务”全维保障网,兑现50年温暖承诺
